The Photoelectric Effect: Light As A Particle Video
A simple, easy-to-understand explanation of the experiment that proved the particle nature of light and won Einstein a nobel prize. The Photoelectric Effect.
Hi! I'm Jade. I make fun physics videos that will make you smarter while making you smile :)
***SUBSCRIBE***
https://www.youtube.com/c/upandatom
***Let's be friends***
TWITTER: https://twitter.com/upndatom?lang=en
***RELATED VIDEOS YOU WILL LOVE***
The Double Slit Experiment: Light as a Wave https://youtu.be/gRX-s0p4HpM
How Many Dimensions Can You See? https://www.youtube.com/watch?v=cwWbSVzAFLQ
Thanks for watching!
____________________________________________________________________
The Photoelectric effect proved the quantum nature of light and won Einstein a nobel prize. It proved that light is made of particles! This discovery was controversial at the time as Thomas Young proved that light was a wave in 1801 with his Double Slit Experiment.
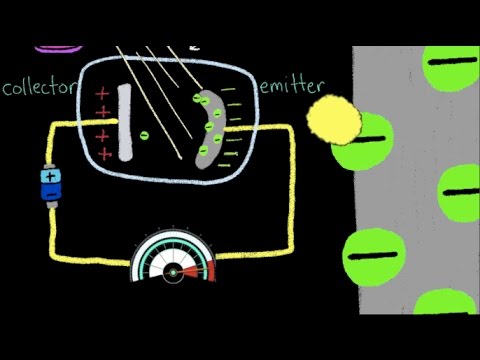
The experiment that proved the quantum nature of light (light is a particle) went like this: A negatively charged metal plate which is called the "emitter" is placed opposite a positively charged metal plate called the collector. They are both placed in a vacuum chamber and connected by a wire and battery. The electrons in the emitter want to jump across to the collector plate, but the vacuum is preventing movement within the chamber. A light source is introduced to give the electrons the energy they need to jump the gap to the collector. The energy from the light is transferred to the electrons in the form of kinetic energy. This flow of electrons produces a current which is measured by an ammeter in the circuit.
If light were a wave, the amount of electrons being freed from the emitter plate should depend on the intensity of the light, not the frequency. Light of higher intensity means a wave of bigger amplitude which means a wave carrying more energy. So a higher intensity should produce a larger current. So if the intensity of the light is high enough, regardless of the frequency of the light, the electrons should be given enough energy to break free. Also, this energy transfer should be gradual as there is a build up of energy,, so there would be a time delay between when the light hit the electrons and when they are emitted. Furthermore, the kinetic energy of the electrons should increase as intensity increases. This means they travel with more speed toward the collector.
Turns out none of that happened. The electrons jumped off the emitter instantaneously, there was no gradual build up of energy. It was as if the electrons were getting knocked off the surface. But the most interesting observation was that the kinetic energy of the electrons was dependent on the frequency of the light, not the intensity. For some frequencies, no electrons jumped off the metal at all. The only way to explain this behaviour is that light is a particle (photon). The threshold frequency (sometimes called stopping potential) is the minimum frequency needed to knock the electrons off the metal surface. All frequencies above the threshold frequency carry enough energy to knock the electrons off the emitter, all frequencies below do not. The higher the frequency of the photons, the more kinetic energy in the electrons.
From this, Einstein concluded that the energy carried in one of these light particles, which he called photons, was also proprtional to the frequency of the light (E=hf). More energy in a single photon meant more energy being transferred to an electron.
The one thing that did depend on the intensity was the electron current. If the light shone was above the threshold frequency, a higher intensity means more photons. More photons means more photon electron interactions which means more emitted electrons.
Einsteins discovery of photons and the photoelectric effect won him the nobel prize. But Young's proof of light being a wave was not wrong. Light is a particle and it's a wave. This is called wave-particle duality and is widely accepted in quantum physics.
About the Site 🌐
This site provides links to random videos hosted at YouTube, with the emphasis on random. 🎥
Origins of the Idea 🌱
The original idea for this site stemmed from the need to benchmark the popularity of a video against the general population of YouTube videos. 🧠
Challenges Faced 🤔
Obtaining a large sample of videos was crucial for accurate ranking, but YouTube lacks a direct method to gather random video IDs.
Even searching for random strings on YouTube doesn't yield truly random results, complicating the process further. 🔍
Creating Truly Random Links 🛠️
The YouTube API offers additional functions enabling the discovery of more random videos. Through inventive techniques and a touch of space-time manipulation, we've achieved a process yielding nearly 100% random links to YouTube videos.
About YouTube 📺
YouTube, an American video-sharing website based in San Bruno, California, offers a diverse range of user-generated and corporate media content. 🌟
Content and Users 🎵
Users can upload, view, rate, share, and comment on videos, with content spanning video clips, music videos, live streams, and more.
While most content is uploaded by individuals, media corporations like CBS and the BBC also contribute. Unregistered users can watch videos, while registered users enjoy additional privileges such as uploading unlimited videos and adding comments.
Monetization and Impact 🤑
YouTube and creators earn revenue through Google AdSense, with most videos free to view. Premium channels and subscription services like YouTube Music and YouTube Premium offer ad-free streaming.
As of February 2017, over 400 hours of content were uploaded to YouTube every minute, with the site ranking as the second-most popular globally. By May 2019, this figure exceeded 500 hours per minute. 📈
List of ours generators⚡
Random YouTube Videos Generator
Random Film and Animation Video Generator
Random Autos and Vehicles Video Generator
Random Pets and Animals Video Generator
Random Travel and Events Video Generator
Random People and Blogs Video Generator
Random Entertainment Video Generator
Random News and Politics Video Generator
Random Howto and Style Video Generator
Random Education Video Generator